News

Contaminants in Textile Recycling: Understanding Their Fate, Detection and Elimination
By UJEP
Author: Jan Snow, Faculty of Environment.
March 2026
Like most synthetic materials, textiles are complex systems. To meet our expectations for durability, color, stretch, water resistance, and comfort, fabrics contain a wide range of polymers, additives, dyes, finishes, and processing agents. While this chemical diversity enables performance and design, it becomes a major obstacle at the end of a product’s life. Today, only about 1% of textiles in the EU are recycled in a true “T-shirt-to-T-shirt” loop [1], and one of the key reasons is contamination.
Textile contaminants range from relatively harmless degraded UV stabilizers, dyes, and pigments to well-known hazardous substances such as phthalates (plasticizers) or PFAS (used for water repellency). In addition to intentionally added additives, textiles may accumulate impurities during use (detergents, oils, environmental pollutants), and further contamination can occur during collection and recycling. From a technological perspective, even materials incompatible with a given recycling route may act as contaminants, for example, the presence of PET in pyrolysis may lead to process fouling due to the formation of solid products.
Careful sorting is therefore a fundamental step in any textile recycling system. However, sorting alone is not sufficient; a thorough understanding of the fate of contaminants in different recycling processes is also essential.
Tracking Contaminants Through Recycling Technologies
Let us now take a closer look at what may happen to contaminants within different recycling processes. The schematic below illustrates an idealized scenario assuming maximum process efficiency. In practice, such perfect separation is rarely achieved. Consequently, contaminants may persist in final products, not only in mechanical recycling, as highlighted in the scheme, but also in chemical recycling pathways.
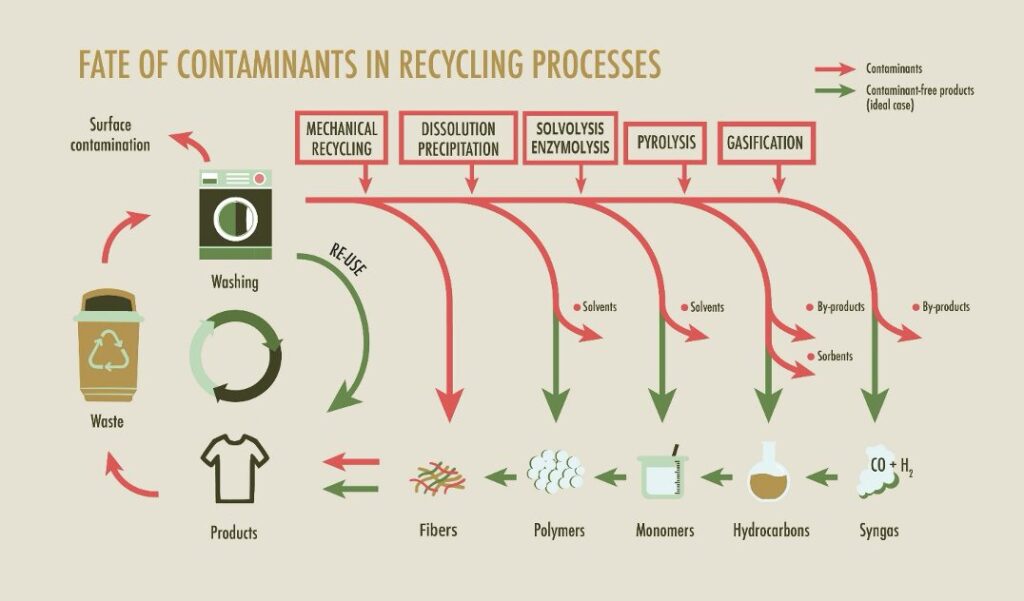
Mechanical Recycling
In mechanical recycling, textiles are shredded and—if synthetic—melted and re-extruded. The possibilities for contaminant removal are limited. During melt processing, the polymer passes through hot filtration systems that capture solid particles such as metal fragments or inorganic pigments. However, molecularly dispersed organic additives, such as plasticizers and UV stabilizers, cannot be removed by simple filtration.
This explains why mechanically recycled materials often exhibit inferior properties and lower added value (downcycling). Some limitations can be mitigated by blending recycled material with virgin polymer or directing products toward open-loop applications. In practice, contaminant control largely depends on ensuring that unwanted substances are not present in the input stream, or, in some cases, on dilution below regulatory thresholds.
Solvent-Based and Depolymerization Processes
Processes based on dissolution, solvolysis, or enzymatic depolymerization offer greater flexibility in terms of contaminant removal. These approaches involve dissolving the polymer and re-precipitating it, or chemically breaking it down into monomers and intermediate products. Contaminants may be separated based on solubility differences, adsorption onto sorbents, filtration of insoluble fractions, or purification of monomers via crystallization or distillation.
Some contaminants may undergo chemical transformation during these processes. While this can reduce their concentration, it may also generate new species with altered toxicity. Purification strategies must therefore be carefully designed and validated.
Pyrolysis
Moving toward thermochemical conversion processes, we encounter pyrolysis. At elevated temperatures (typically 350–500 °C) in an oxygen-free environment, polymers decompose into liquid hydrocarbons, gases, and solid residue.
A key advantage of pyrolysis is that many inorganic contaminants, such as metals and mineral pigments—remain in the solid residue. Some organic additives decompose, while others may form new compounds present in the pyrolysis oil. The high temperature opens the door for extensive chemical transformations, and catalysts or sorbents can support contaminant capture. The distribution of contaminant-containing fractions can also be influenced by carefully controlling the temperature profile.
Gasification
At even higher temperatures, gasification converts materials into synthesis gas (CO and H₂). This route is generally considered a last resort for materials unsuitable for other treatments. In gasification, most organic contaminants are destroyed due to the extreme conditions, while inorganic elements are concentrated in ash or slag. Although contaminants are less problematic in the final product stream, gas cleaning systems are still required.
Where Should We Monitor Contaminants?
Contaminants may need to be monitored at several stages:
- Input materials – collected textile waste
- Main products – recycled polymers, purified monomers, or pyrolysis oils
- By-products and waste streams – washing water, spent solvents, filtration residues, gaseous emissions, and solid residues
A mass-balance perspective is crucial to understand, quantify, and control how contaminants redistribute across process streams.
Detection: Laboratory vs. Industrial Context
Contaminants occur in solids, liquids, and gases, requiring a broad analytical toolbox. It is also important to distinguish between laboratory-scale methods and techniques feasible in industrial practice.
If we start from the final products—ideally, for example, a new T-shirt—at the Faculty of Environment, UJEP, we apply ICP-MS and XRF/XRD to determine elemental composition. For the identification of specific organic additives, we employ pyrolysis–gas chromatography–mass spectrometry (Py-GC/MS), which provides detailed molecular information directly from solid materials.
A limitation of ICP-MS and Py-GC/MS is that they require relatively small sample quantities, which is the trade-off for their high sensitivity. As an alternative, we can perform pyrolysis on a larger scale (0.5–1 kg) and analyze the resulting liquid fraction using comprehensive two-dimensional GC-MS to obtain characteristic chemical fingerprints of target contaminants. Gas and liquid chromatography are also used to analyze solvolytic products, post-process solvents, and other secondary streams.
If textile recycling is to be implemented at a larger scale, we must guarantee the safety and compliance of recycled products accordingly. There is still considerable room for the development of new analytical methods and evaluation approaches. XRF currently appears to be one of the most robust techniques, as it can be applied directly in sorting facilities. However, its lower sensitivity and the heterogeneity of textile materials remain significant challenges.
Moving Forward
Many advanced textile recycling technologies are still under development. Although laboratory studies demonstrate that contaminants can often be removed or transformed, scaling these solutions requires technical optimization, economic viability, and regulatory compliance.
I wish success to all project partners and everyone working in textile recycling—and to all of us who hope to wear high-quality, safe, and environmentally friendly clothes.
[1] European Parliament, The impact of textile production and waste on the environment, European Parliament (2020). https://www.europarl.europa.eu/topics/en/article/20201208STO93327/the-impact-of-textile-production-and-waste-on-the-environment-infographics
